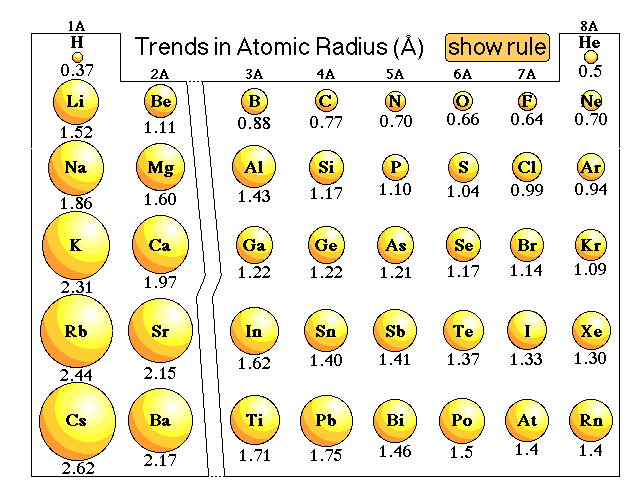
The protons in the nucleus attract the valence electrons in the outer energy level because of opposite electrostatic charges. One other contributing factor to atomic size is something called the shielding effect. It becomes apparent that as we move downward through a family of elements, that each new atom has added another energy level and will, therefore, be larger. The Periodic Table from period to period, each successive period represents the addition of a larger energy level. Energy level 2 is larger than energy level 1, energy level 3 is larger than energy level 2, and so on. When we first learned about principal energy levels, we learned that each new energy level was larger than the one before. Each row (period) in the periodic table represents another added energy level. Each atom in this family (and all other main group families) has the same number of electrons in the outer energy level as all the other atoms of that family. Let's now look at the atomic radii or the size of the atom from the top of a family or group to the bottom. The size of the atom is slightly larger, approximately 2 ×10 −8 cm in diameter.Ītomic Size in a Column Increases from Top to Bottom He was able, using technology available in the early part of the 1900s, to determine quantitatively that the nucleus had an approximate size of 4 ×10 −12 cm. The gold foil experiment by Rutherford, first done in 1911, is of particular interest to us in this unit because it was this experiment that first gave science an approximate measurement for the size of the atom. How do we measure the size of the atom? Ernest Rutherford is famous for his experiments bombarding gold foil with alpha particles. The measurement would be taken as one-half the distance between the nuclei of the hydrogen atoms in a diatomic hydrogen molecule. The figure below represents a visualization of the atomic size definition.Ī visual representation of the atomic radius of a hydrogen atom. The atomic radius is one-half the distance between the centers of a homonuclear diatomic molecule (a diatomic molecule means a molecule made of exactly two atoms and homonuclear means both atoms are the same element). One way that chemists define atomic size is by using the atomic radius. Atomic size is defined in several different ways and these different definitions often produce some variations in the measurement of atomic sizes.īecause it is so difficult to measure atomic size from the nucleus to the outermost edge of the electron cloud, chemists use other approaches to get consistent measurements of atomic sizes. The region in space occupied by the electron cloud of an atom is often thought of as a probability distribution of the electrons and therefore, there is no well-defined "outer edge" of the electron cloud. due to complex structure of f-orbital and poor shielding of f-e(s)= lanthanoids contractions occur.Ītoms Have No Definite Boundary.Use the concept of effective nuclear charge to explain why the atomic radii of the main group elements increase when we move down a group in the periodic table.Use the general trends to predict the relative sizes of atoms.Describe how the trend of atomic radii works for transition metals.Describe the trend of atomic radii in the rows in the Periodic Table.Describe the general trend in atomic size for groups and for periods.Describe the factors that determine the trend of atomic size.Describe measurement methods for atomic size.State the boundary issue with atomic size.The number of electrons held between the nucleus and its outermost electrons (called the shielding effect).The number of energy levels holding electrons (and the number of electrons in the outer energy level).The number of protons in the nucleus (called the nuclear charge).The actual trends that are observed with atomic size have to do with three factors. Each of these trends can be understood in terms of the electron configuration of the atoms. The two following this lesson will discuss ionization energy and electron affinity. The first lesson of this chapter is devoted to the trend in atomic size in the Periodic Table. There are three specific periodic trends that we will discuss. This means is that as you move down a group or across a period, you will see a trend-like variation in the properties. In the Periodic Table, there are a number of physical properties that are not really "similar" as it was previously defined, but are more trend-like. 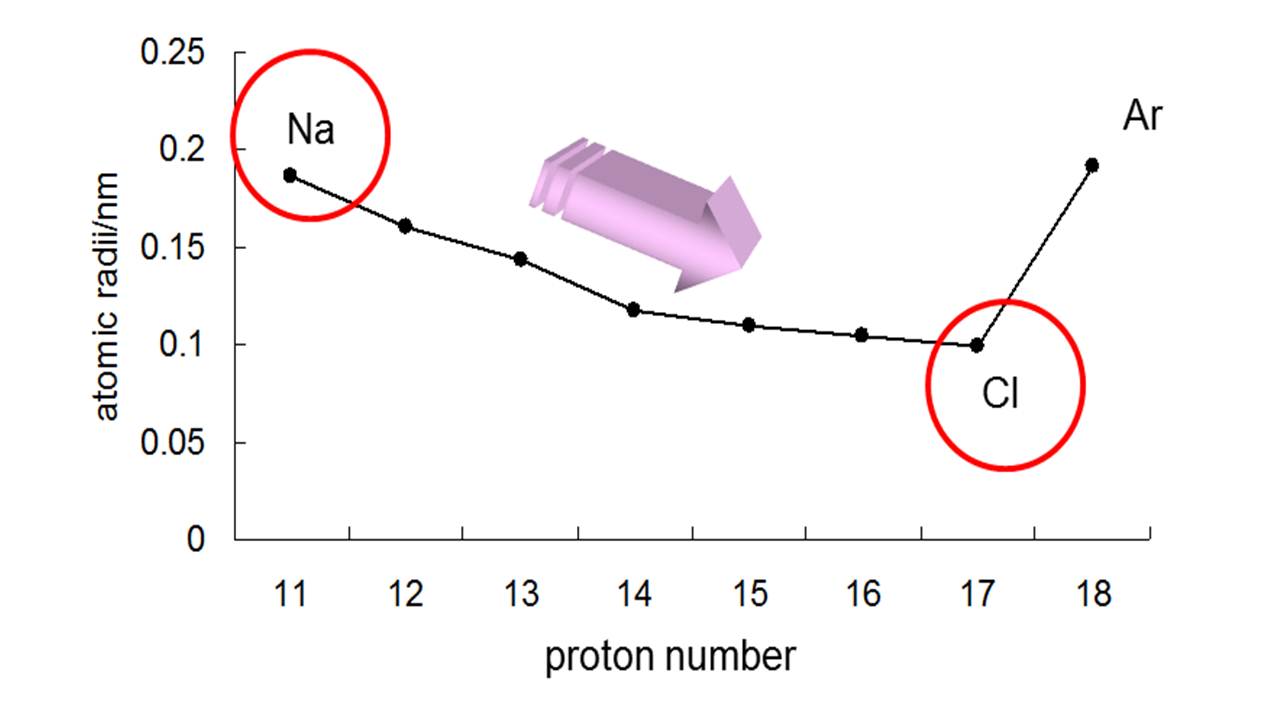 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
